New Utrasonic Medical Applications Based on Acoustically Induced Microbubbles
Researchers continue to develop innovative new medical techniques and applications for sound waves: specifically, those in the ultrasonic region. First introduced in 1942 by the Austrian scientist Karl Theo Dussik, ultrasonic imaging remains one of the most reliable, safe, and simple imaging techniques in medicine. Yet today, ultrasound is being used for so much more than basic medical imaging.At greater intensities, focused ultrasonic pulses are now used to break up and liquefy body tissue: disintegrating gall bladder stones, or breaking down tumors in the brain and pancreas, for example. And in the last 20 years, therapeutic ultrasound has been explored for targeted drug delivery, to cauterize wounds, and even disrupting bacterial biofilms.
One of the key mechanisms for many of these useful effects appears to be microbubbles, induced by acoustic cavitation. “Recent studies are showing that microbubbles can help us address a variety of current clinical needs,” Vesna Zderic (George Washington University) reported at last fall’s meeting of the Acoustical Society of America (ASA).
 Photo: George Lewis Jr A therapeutic ultrasound transducer exerting a force on the surface of water. |
Miniscalpels and Biofilms. It may one day be possible to exploit acoustic cavitation to make surgical incisions inside the body with no need to open or puncture the skin in any way, according to Zhen Xu of the University of Michigan. He and his colleagues are investigating the effects of applying high-intensity ultrasound pulses focused into tiny acoustic “scalpels,” to test whether they can deliver acoustic power without heating to tissues deep within the body.
The high-intensity ultrasound causes microbubbles to form at the focal point. These bubbles expand and collapse with great force–cavitation–and this activity can mechanically fragment tissues. Xu believes this may occur because cell membranes cannot withstand the pressure caused by the bubbles.
Xu’s team has succeeded in focusing acoustical beams into a cluster of “miniscalpels” roughly the size of a single cell. They can control the beams electronically using a computer mouse or a joystick. Also, the surgery can be precisely targeted and monitored in real time because the microbubbles are easily tracked with conventional ultrasound or MRI technologies.
Microbubbles can also help break up biofilms, the protective slicks formed by single-celled organisms when they clump together on a solid surface, communicate with each other, and secrete a mucus-like substance. Such films can form inside pipes and pollute water supplies, or create a slippery slime on creek rocks. Bacterial films like Staph aureus can form on medical devices in the human body, causing severe infection, and even death. These mucus secretions protect the bacteria in biofilms from being easily killed.
E. Carr Everbach and his colleagues at Swarthmore College recently demonstrated that acoustical waves can disrupt biofilms and destroy bacteria. They grew biofilms of a strain of fluorescing E. coli on microscope slides sandwiched between two piezoelectric bars. They placed ultrasound transducers directly onto the microscope slide to avoid shifting the placement of the samples during the laser zapping process.
All these things enabled them to view the biofilm under a confocal microscope before, during, and after charging the piezoelectric bars, thereby setting up standing acoustical waves on the biofilms. They observed changes in the biofilm structure that showed a mechanical destruction of the film and bacteria. Specifically, the dead bacteria didn’t fluoresce. Once a chunk of biofilm has broken off, it is easier to kill the bacteria with antibiotics.
Microbubbles, they found, were key. “If you do this without the presence of any bubbles, not much happens,” said Everbach. “What causes the disruption of the bacteria isn’t the ultrasound directly, but microscopic bubbles that we introduce into the system, which are directed by the ultrasound into acoustic cavitation.” Bubbles can also form naturally as part of the biological metabolism of bacteria. “We’re trying to understand how much we even need external bubbles,” he said. “We can look at the interaction of bubbles, ultrasound, and cells under a microscope in real time to get a better idea of what’s going on.”
Idea for Using Ultrasound
George Lewis, Jr. got the idea of using ultrasound for targeted anticancer drug delivery from an Indian study on using sono-poration for transdermal drug delivery. This is an older technique in which the drug is applied to the skin, and then ultrasound is applied which breaks down the skin surface so the drug can better permeate through. The Indian leather industry uses a similar technique to help dyes diffuse into the leather, resulting in a more uniform color.
George Lewis, Jr. got the idea of using ultrasound for targeted anticancer drug delivery from an Indian study on using sono-poration for transdermal drug delivery. This is an older technique in which the drug is applied to the skin, and then ultrasound is applied which breaks down the skin surface so the drug can better permeate through. The Indian leather industry uses a similar technique to help dyes diffuse into the leather, resulting in a more uniform color.
The research is still in the early stages, but Everbach believes the technique could be applied in personalized water sterilization systems, and to fight hospital-acquired infections more effectively, particularly by curtailing the buildup of biofilms on implanted medical devices. “We’re interested in understanding the fundamental mechanisms of interaction, not jumping right to some industrial scrubbing process.”
“Rubbing the Brain.” Neurosurgeons can usually successfully remove as much as 99.5% of a brain tumor when they operate, but they can't be as aggressive about removal as they might be in other, less sensitive areas of the body. A few scattered cancer cells are usually left over, which are treated with powerful anti-cancer drugs.
Recent progress in this area includes the development of “gliodel wafers”: disc-shaped implants infused with cancer-fighting drugs that are placed at the site where a tumor used to be just before the neurosurgeon closes everything up after removing a brain tumor. This means the drugs can dissolve and diffuse slowly into the surrounding brain tissue to kill any lingering cancer cells. However, pharmaceutical agents don’t appear to penetrate brain tissue uniformly–something that puzzles researchers.
Brain cancers are especially challenging, according to George Lewis Jr., a researcher at Cornell’s department of biomedical engineering. Some of the newer drugs can easily kill any straggling cancer cells–provided the drug can reach them. Cancer cells migrate to other areas of the brain rather quickly after surgery. Sure, it's only a few millimeters to a centimeter, but it's just enough to elude the drugs. “In two weeks you have tumors reappearing, and in two months, the patient is dead,” Lewis says. That’s why brain cancers like neuroblastomas and neurofibromatosis are still the leading cause of cancer-related death in people under the age of 35.
Lewis is investigating the use of acoustic pulses to help brain tissue absorb chemotherapy drugs faster–before the cancer cells have a chance to migrate very far–and also increase the range of diffusion. He and his collaborators at Yale and Princeton use focused ultrasound to agitate the tissue matrices, enhancing permeability and making it easier for the drug to get into the brain tissue. Basically, they’re massaging the brain tissue to open up the pores, since the brain is kind of similar to a sponge.
Initial results from experiments with a horse brain indicate that with such a technique, the drugs do indeed spread further and faster into the tissue than they would by natural diffusion alone–a hundredfold further, in fact, which makes it very promising for future treatment of brain cancers. They’re now carrying out a full study using live animals to see if they still get enhanced diffusion effects, and also to make sure a living creature can withstand the treatment.
It is still not entirely clear what mechanism is actually at work in the technique. Some of Lewis’s collaborators suspect that acoustic cavitation from microbubbles works to bloat the pores and open them up sufficiently so the drugs can diffuse through the tissue more effectively. Lewis thinks it might be primarily a mechanical effect related to the acoustic waves: “They go through the tissue as a compression wave, which oscillates the tissue and massages it to allow the drug more readily to diffuse through it.” He likens it to how dentists will often massage a patient’s gum when injecting Novacaine. “We’re trying to rub the brain” using ultrasonic waves.
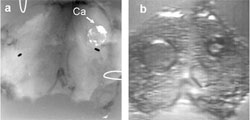 Photo: Azra Alizad a) X-ray and (b) vibro-acoustography (VA) images of an excised human thyroid. The VA image shows a calcified lesion on the right side and a cyst on the left side of the thyroid. |
Alizad and his colleagues have developed a novel non-invasive imaging technique called vibro-acoustography (VA) that is especially sensitive to tissue stiffness, and also produces high resolution and high contrast images. The technique employs ultrasound to vibrate thyroid tissue at low frequencies, and the resulting vibrations are detected by a highly sensitive microphone. According to Alizad, harder tissue produces a significantly different acoustic field than softer tissue, so it is easier to detect the difference between them.
Most recently, Alizad has tested the technique on excised human thyroids from autopsy, and found that VA images showed calcifications, anatomic details, tissue structures, and nodules when present. He has yet to test the technique in clinical trials, but VA is currently being clinically evaluated for the detection of breast cancer lesions in human trials.
Medical acoustics has come a long way since doctors first used stethoscopes to listen to the human heart at the dawn of the 19th century. No doubt scientists will develop even more advanced applications of focused acoustical energy for the medical field in the future.
©1995 - 2024, AMERICAN PHYSICAL SOCIETY
APS encourages the redistribution of the materials included in this newspaper provided that attribution to the source is noted and the materials are not truncated or changed.
Contributing Editor: Jennifer Ouellette
Staff Writer: Ernie Tretkoff
April 2008 (Volume 17, Number 4)
Articles in this Issue

