Einstein and The Photoelectric Effect
Editor's Note: With the January 2005 issue of APS News, we begin a year-long series of columns devoted to the work and legacy of Albert Einstein-our humble contribution to the World Year of Physics. Columns will appear in chronological order of Einstein's work, regardless of the month associated with the topic at hand.
 Einstein in the Patent Office years. 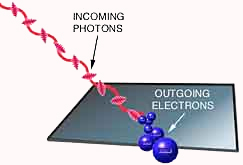 Illustration Credit Einstein noted that careful experiments involving the photoelectric effect could show whether light consistes of particles or waves. |
What was puzzling was that different metals required bursts of different minimum frequencies of light for the electron emission to occur, while increasing the brightness of the light produced more electrons, without increasing their energy. And increasing the frequency of the light produced electrons with higher energies, but without increasing the number produced. This became known as the photoelectric effect, and it would be understood in 1905 by a young scientist named Albert Einstein.
Einstein's fascination with science began when he was 4 or 5, and first saw a magnetic compass. He was enthralled by the invisible force that caused the needle to always point north, and the instrument convinced him that there had to be "something behind things, something deeply hidden." He spent the rest of his life trying to decipher the arcane mysteries of the universe.
Today, the name Einstein is synonymous with genius, but for years his parents thought their son was a bit "slow" because he spoke hesitantly and wasn't a stellar student. Einstein was just plain bored with the rote teaching methods of formal education, with its emphasis on memorization and blind obedience to an arbitrary authority. He preferred to study at home with books on math, physics and philosophy. "It's almost a miracle that modern teaching methods have not yet entirely strangled the holy curiosity of inquiry," he later said. "For what this delicate little plant needs more than anything, besides stimulation, is freedom."
Einstein found both when he attended a local Swiss school in Aarau, having failed the entrance exam for the more prestigious Swiss Federal Institute of Technology. For the first time, he had teachers who gave him the freedom and latitude to pursue his own ideas, and he threw himself into studying the electromagnetism theories of James Clerk Maxwell, which were rarely taught at universities at the turn of the century. Then he studied physics at the Institute of Technology in Zurich, but graduated with an undistinguished academic record, and failed to obtain a university post teaching mathematics and physics. Instead, he worked as a patent clerk in Bern, doing theoretical physics on the side, and occasionally meeting with a group of friends to read and discuss books on science. They called themselves the "Olympia Academy."
In March 1905, Einstein—still a lowly patent clerk in Switzerland—published a paper explaining the photoelectric effect. Five years earlier, Max Planck had solved the problem of black body radiation by showing that each atom making up the walls of the cavity could only absorb or emit radiation in discrete "quanta" such that the energy of each quantum is an integer multiple of its frequency times a new fundamental constant.Planck thought his concept of quanta was just a mathematical "trick" to get theory to match experiment. But Einstein extended Planck's quanta to light itself. (Planck had assumed that just the vibrations of the atoms were quantized.) Light, Einstein said, is a beam of particles whose energies are related to their frequencies according to Planck's formula. When that beam is directed at a metal, the photons collide with the atoms. If a photon's frequency is sufficient to knock off an electron, the collision produces the photoelectric effect. As a particle, light carries energy proportional to the frequency of the wave; as a wave it has a frequency determined by the particle's energy. Einstein won the 1921 Nobel Prize in physics for this work. But it was just the beginning.
Next month: special relativity.
See an online exhibit about Albert Einstein by the American Institute of Physics at http://www.aip.org/history/einstein/.
©1995 - 2024, AMERICAN PHYSICAL SOCIETY
APS encourages the redistribution of the materials included in this newspaper provided that attribution to the source is noted and the materials are not truncated or changed.
Associate Editor: Jennifer Ouellette

