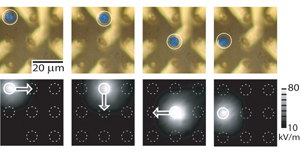
 To appear in article by T.P. Hunt, H. Lee, and R.M. Westervelt in Applied Physics LetterSingle cell manipulation. Top: microscope pictures of yeast cell (circled, high-lighted in green) following a trap that moves in a clockwise direction. Bottom: simulations of electric trapping field. Arrows show direction of trap movement. |
Harvard University's Hakho Lee has built a microelectromagnetic matrix of wires and insulators capable of trapping and moving a cell continuously in a fluid to position cells at desired locations. Being able to hold and move a single cell is a very important capability in biological research, and is usually achieved using fluidic or optical (laser) methods.
Lee's device builds on what biologists already do: manipulate cells by using magnetic beads, a chemically modified surface that can be specifically bound to a target biological system. This bead-bound sample can be manipulated with an external magnetic field, permitting fast and easy control of the sample.
Lee's matrix has two arrays of straight wires aligned perpendicular to each other. Each wire is connected to a separate computer-controlled current source, and the current in each wire is adjusted to generate optimized magnetic field patterns for various experimental purposes.The matrix can trap a single yeast cell, for example, and move it to a desired location, move multiple cells along different paths simultaneously, or rotate or twist cells to study their mechanical properties. Simultaneous and independent control of multiple cells is Lee's ultimate goal, since this would enable bioassisted assembly nanomagnets or nanoparticles.
Lee's Harvard colleague, Tom Hunt, has adopted a somewhat different approach, using electricity to manipulate and move cells. He built a micropost matrix array of electrodes, each of which is connected to a computer-controlled voltage supply. Running a simple computer program sets up non-uniform electrical fields that were shown to move cells or particles.
Even though cells are electrically neutral, they develop small charge separations when they are placed in an electrical field (polarization). The field exerts a force on the polarized cell, trapping it without physically touching or damaging the cell. By generating the proper electric field, almost any particle, including cells and DNA molecules, can be trapped and moved without any need for special sample preparation.
Lee's and Hunt's respective matrices could one day be used to make a reprogrammable microfluidic system. Several different experiments could be performed with one generic microfluidic chamber, simply by controlling the motion of cells—not by the physical barrier of the chamber walls, but rather by electric or magnetic fields that can be dynamically reconfigured for different experiments.
In the future, advanced versions of these matrices could hold thousands of both cancerous and normal cells in individual traps. Dozens of slightly different chemotherapy drugs would wash over the cells, and the embedded sensors in the array would keep track of how many cells survived each treatment, enabling scientists to rapidly test the effects of new potential drug compounds.
In a new experiment conducted at Bell Labs/Lucent Technologies, a liquid drop was maneuvered around a special surface consisting, at the microscopic level, of a forest of tiny stalks. The blades of this "nanograss" can be selectively electrified so as to move the drop from place to place, or to cause it to lose its spherical shape and wet the surface below. The result is a "tunable" surface, according to Lucent scientist Tom Krupenkin.
The conversion of the surface from being hydrophobic (the drop staying aloof at the top of the blades) to hydrophilic (the drop collapsing and flooding the plain between the blades) could result in many potential applications.
Heat mitigation is one example. Drops could be delivered to hot spots on microchips, where the drop could douse the troubled area, much like airborne drops of water can help douse a forest fire. It would absorb the heat, and then depart. Optical properties of the surface could also be switched from one state to another through electronically controlled wetting.
Potential microfluidics applications include combinatorial chemistry in microreactors, drag reduction, or altering the friction of channels. In microbatteries, electrochemicals could be kept isolated until energy was actually needed, thus extending the battery's working life and saving energy for moments of peak activity.
Other interesting nanodevices described at the meeting included the world's smallest synthetic motor, developed by Lawrence Berkeley Laboratory's Adam Fennimore.
The nanomotor uses electric instead of magnetic fields to attract the rotor to different positions, and uses nested cylinders of multi-walled carbon nanotubes to build the rotational bearing. One potential application would be its use as a full-control rotational mirror in optical switching for the telecommunications, although it could be used anywhere in MEMS devices where one needs rotational freedom.
Ian Chan of the University of California, Berkeley, has used an array of artificial micropores mounted on a chip to selectively identify many types of analytes simultaneously. This technique achieves biological sensing by detecting changes in the size of colloids, since the height of the pulse depends upon the size of the colloids. That size changes with the presence of a specific antigen to which the colloid is sensitive.
Chan hopes to extend the concept to multianalyte detection by adding different-sized colloids that are sensitive to different antigens, or by using up to six different multiple pores at one time.
©1995 - 2024, AMERICAN PHYSICAL SOCIETY
APS encourages the redistribution of the materials included in this newspaper provided that attribution to the source is noted and the materials are not truncated or changed.
Associate Editor: Jennifer Ouellette
May 2004 (Volume 13, Number 5)
Articles in this Issue

