 Dynamic Holographic Optical Tweezers 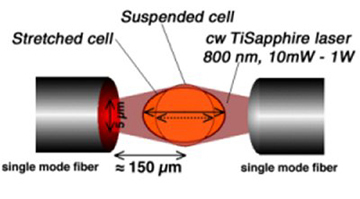 |
Carl Hansen of CalTech has devised the most complex microfluidic testbed to date, boasting thousands of micromechanical valves and hundreds of addressable chambers. His device has the largest degree of integration yet achieved: a chip with 1000 250-picoliter chambers with attendant valves for controlling flow and mixing. This makes it ideal for large- batch processing of protein crystal growth and other biomolecule studies.
Another device in the Caltech lab of Hansen's colleague, Stephen Quake, allows the careful metering of reagents in order to facilitate protein crystallization under a variety of conditions, such as pH, viscosity, surface tension, or various different solvents. The device is capable of producing 144 parallel reactions, which can take place using only 10nl of precious protein samples—100 times less than with usual methods. In this way, many proteins have been transformed into crystals, often in the space of hours rather than days, and some species were crystallized for the first time. The crystals can then be bombarded with x- rays in order to determine molecular structure.
The University of Chicago's David Grier has created multiple optical tweezers using holography, in which a beam of laser light, sent into a hologram, is divided into a myriad of sub-beams, which can independently suspend and manipulate numerous tiny objects for possible transportation, mixing or reacting. Grier showed movies of ensembles of microspheres moved into patterns and set to spinning by the holographically sculpted light fields.
Applied to fluid samples of biomolecules, the holographic multiplexing produces what Grier calls "optical fractionation," an optical equivalent of gel electro- phoresis, in which electric fields are used differentially to drive and separate macromolecules. In the more flexible Chicago approach, there is no viscous gel, and a deft change in the computer generated hologram or the laser wavelength can quickly bring about sorting of objects ranging from the 100-nm size (the scale size of viruses, for example) up to the 100-micron size scale.
Jochen Guck of the University of Leipzig in Germany has invented an "optical stretcher," a device in which cells moving through fluid channels are sorted and studied by squeezing the cells. Since sick cells are softer (by a factor of 2 to 10) than healthy cells, the process can differentiate between healthy and sick cells, at a rate of hundreds of cells per hour, compared to typical rates of 10 cells per day using other elastisizing methods, thus reducing the need for biopsies requiring larger tissue samples. The device might even be able to differentiate between ordinary cancer cells and mestasizing cancer cells, which are even softer. Guck subjects fluid-borne cells to a pair of laser beams which stretch the cells and probe their elasticity.
©1995 - 2024, AMERICAN PHYSICAL SOCIETY
APS encourages the redistribution of the materials included in this newspaper provided that attribution to the source is noted and the materials are not truncated or changed.
Associate Editor: Jennifer Ouellette
May 2003 (Volume 12, Number 5)
Articles in this Issue

